
Chemistry, 04.02.2021 04:50, aliviadushane
How many moles of carbon dioxide are produced from the complete combustion of 5.42 moles of ethanol? 2C2H6O +7 O2 ®4 CO2 + 6H2O

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:10, jojomgarcia01
When water dissociates, each water molecule splits into a hydroxide ion and a) h 3 o + b) a hydrogen atom c) a hydrogen ion d) h 2 o e) oh —
Answers: 2

Chemistry, 22.06.2019 11:00, coco8560
Freezing and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1


Chemistry, 22.06.2019 20:00, Chynadoll94
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1
Do you know the correct answer?
How many moles of carbon dioxide are produced from the complete combustion of 5.42 moles of ethanol?...
Questions in other subjects:

English, 23.06.2019 03:10

History, 23.06.2019 03:10


Social Studies, 23.06.2019 03:10

Chemistry, 23.06.2019 03:10


Mathematics, 23.06.2019 03:10

Mathematics, 23.06.2019 03:10

Mathematics, 23.06.2019 03:10


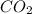 will be produced from complete combustion of 5.42 moles of ethanol
will be produced from complete combustion of 5.42 moles of ethanol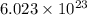 of particles.
of particles.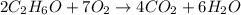
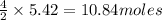 of
of 



