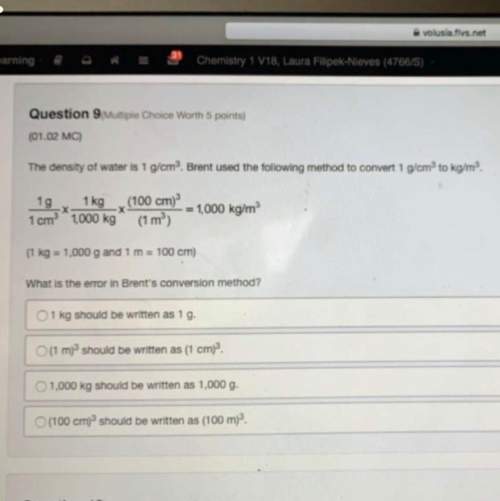
What is the molality of a solution made by dissolving 2 moles of NaOH in 6 kg
of water?
A. 0....

Chemistry, 02.02.2021 20:40, itscheesycheedar
What is the molality of a solution made by dissolving 2 moles of NaOH in 6 kg
of water?
A. 0.33 mol/kg
B. 12 mol/kg
C. 3 mol/kg
D. 2 mol/kg

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 04:00, speris1443
Write the balanced equation for a reaction between aqueous nitric acid (hno3) and solid lithium metal (this is a single replacement reaction)
Answers: 1

Do you know the correct answer?
Questions in other subjects:


Mathematics, 08.11.2019 06:31

Chemistry, 08.11.2019 06:31

Mathematics, 08.11.2019 06:31


Mathematics, 08.11.2019 06:31