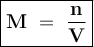Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 06:30, aurikmah2005
Acertain atom has 22 protons and 19 electrons. this atom loses an electron. the net charge on the atom is now 4+1+01-4-. if this same atom with 22 protons and 19 electrons were to gain 3 electrons, the net charge on the atom would be 3+2+02-3-.
Answers: 1

Chemistry, 23.06.2019 10:30, krlx
When a chemist collects hydrogen gas over water, she ends up with a mixture of hydrogen and water vapor in her collecting bottle. if the pressure in the collecting bottle is 97.1 kilopascals and the vapor pressure of the water is 3.2 kilopascals, what is the partial pressure of the hydrogen?
Answers: 1
Do you know the correct answer?
The molecular weight of KMnO4 is 158 g/mol. If you wanted to make 500 mL of a 0.1M stock solution of...
Questions in other subjects:





Physics, 02.03.2020 18:23


Mathematics, 02.03.2020 18:23