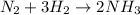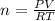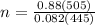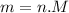Suppose a chemical engineer studying a new catalyst for the Haber reaction finds that liters per second of dinitrogen are consumed when the reaction is run at and the dinitrogen is supplied at . Calculate the rate at which ammonia is being produced. Give your answer in kilograms per second. Be sure your answer has the correct number of significant digits.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:40, yah2muchh
Calculate: select the worksheet tab. this tab you calculate the analyte concentration. fill in the first set of boxes ("moles h2so4" and "moles naoh") based on the coefficients in the balanced equation. (if there is no coefficient, the value is 1.) record the appropriate volumes in the "ml naoh" and "ml h2so4" boxes. record the concentration of the titrant in the m naoh box. click calculate. what is the concentration listed
Answers: 2



Chemistry, 22.06.2019 08:30, aydenmasonc
Which statement describes james chadwick’s discovery.
Answers: 2
Do you know the correct answer?
Suppose a chemical engineer studying a new catalyst for the Haber reaction finds that liters per sec...
Questions in other subjects:

Social Studies, 26.10.2020 01:00

Health, 26.10.2020 01:00


Chemistry, 26.10.2020 01:00


English, 26.10.2020 01:00

Mathematics, 26.10.2020 01:00


Geography, 26.10.2020 01:00

Mathematics, 26.10.2020 01:00