
20
20
RE
lin
DIG
DS
an
10000
na mim
Figure 1
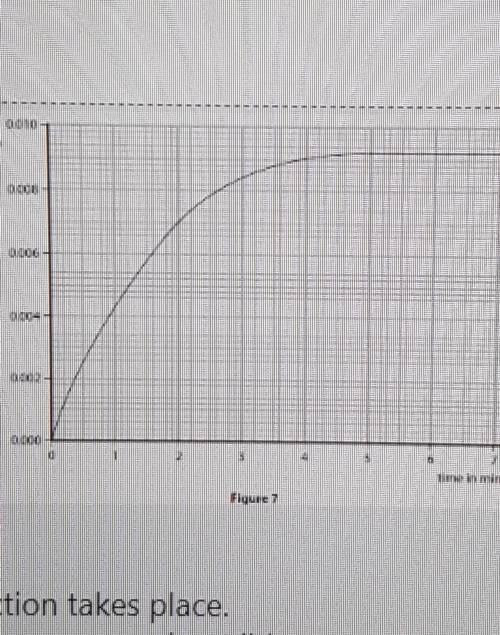
The graph shows that the rate of reaction slows as the reaction takes place.
Explain, in terms of particles, why the rate of reaction between magnesium ribbon and dilute
hydrochloric acid slows as the reaction takes place. M
(3 Points)

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 04:00, nothingworksoutforme
The image shows a process that releases nuclear energy which statement best identifies the process shown the process must be fusion because energy is released the process must be fusion because of have your nucleus formed a smaller nuclei the process must be fission because a large nucleus breaks into smaller nuclei the process must be fission because neutrons are formed
Answers: 1



Chemistry, 22.06.2019 21:00, melissalopez12
Acandle’s wick is the fabric string that holds the flame, and it burns down at a constant slow pace when the candle is lit. the wick is usually surrounded by wax. which is the most important property of covalent compounds that makes them useful for making candle wax? a low boiling point a low melting point a high boiling point a high melting point
Answers: 1
Do you know the correct answer?
20
20
RE
lin
DIG
DS
an
10000
na mim
Figure 1
The graph...
RE
lin
DIG
DS
an
10000
na mim
Figure 1
The graph...
Questions in other subjects:

Advanced Placement (AP), 06.11.2020 21:30


Mathematics, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30

Arts, 06.11.2020 21:30

Mathematics, 06.11.2020 21:30


Mathematics, 06.11.2020 21:30








