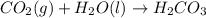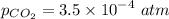Chemistry, 27.01.2021 19:50, exoticbunnylover123
The raw water supply for a community contains 18 mg/L total particulate matter. It is to be treated by addition of 60 mg alum (Al2(SO4)3 14H2O) per liter of water treated. Essentially, all the added alum precipitates represented by the following reaction:
Required:
a. For a total flow of 8000 m^3/d, compute the daily alum requirement and the concentration of solids in the water following alum addition, assuming the alum all precipitates as Al(OH)3(s).
b. The water is initially at pH 7.5 and has ALK=40 mg/L as CaCO3. It is desired to maintain solution pH at 6.5 or higher. Will the pH be in the acceptable range after the chemical addition and Al(OH)3(s) precipitation?
c. What will the pH be if the treated solution is bubbled with air, so that it reached equilibrium with atmospheric CO2?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, viktoria1198zz
Complete the following reactions using word and balanced equations including states. dilute phosphoric acid is added with a calcium hydroxide solution.
Answers: 1


Chemistry, 22.06.2019 05:00, smartboy2296
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 16:00, annsmith66
What statement goes against the kinetic theory of gases
Answers: 1
Do you know the correct answer?
The raw water supply for a community contains 18 mg/L total particulate matter. It is to be treated...
Questions in other subjects:



Social Studies, 18.12.2020 21:30

Mathematics, 18.12.2020 21:30

Mathematics, 18.12.2020 21:30

Chemistry, 18.12.2020 21:30

Mathematics, 18.12.2020 21:30

Mathematics, 18.12.2020 21:30



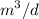
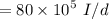
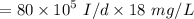
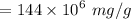
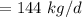
![$ [Al_2(SO_4)_3.14 H_2O] $](/tpl/images/1069/9830/978e1.png) required for one litre of the water treatment.
required for one litre of the water treatment.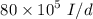

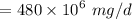
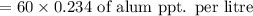
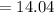 mg of alum ppt. per litre
mg of alum ppt. per litre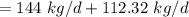
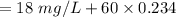
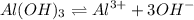
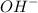 ions.
ions.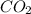 and water as follows :
and water as follows :