
Chemistry, 26.01.2021 22:40, CutiePie8960
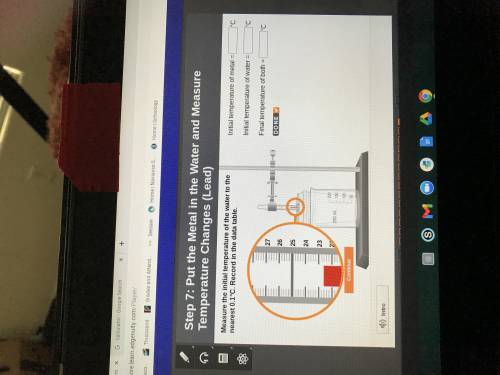
The temperature at 300 seconds (5 minutes) is the
final temperature of both the metal and the water.
Record it to the nearest 0.1°C in the data table.
Initial temperature of metal =
Initial temperature of water = 0
Final temperature of both =
'c
RETRY
250 ml
Continue (60s more)
05:00

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 00:30, catdog5225
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 05:00, adrian128383
What forms when chemical reactions combine pollution with sunlight?
Answers: 1

Chemistry, 22.06.2019 13:00, carlinryan
16. why must the number of electrons lost equal the number of electrons gained in every redox reaction? use 3 – 4 sentences in your own words to address this question. 18. what type of radiation is emitted when chromium-51 decays into manganese-51? show the nuclear equation that leads you to this answer. 19. a radioactive nucleus alpha decays to yield a sodium-24 nucleus in 14.8 hours. what was the identity of the original nucleus? show the nuclear equation that leads you to this answer.
Answers: 2
Do you know the correct answer?
The temperature at 300 seconds (5 minutes) is the
final temperature of both the metal and the water...
Questions in other subjects:




Chemistry, 24.10.2020 04:40

Chemistry, 24.10.2020 04:40


English, 24.10.2020 04:40

Mathematics, 24.10.2020 04:40

Mathematics, 24.10.2020 04:40







