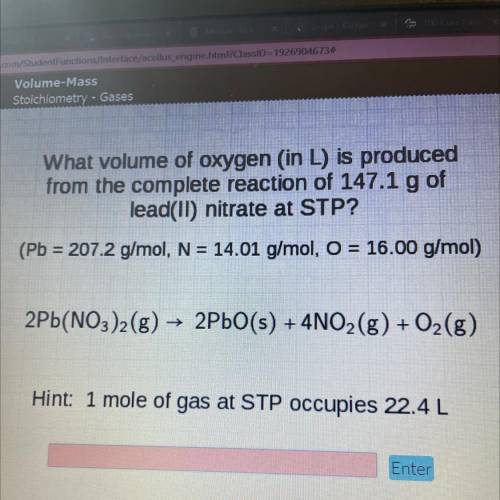
Lus
What volume of oxygen (in L) is produced
from the complete reaction of 147.1 g of
l...


Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:20, juandavidklingera553
What would you do if you told the guy you liked that you liked him
Answers: 1

Chemistry, 22.06.2019 10:10, andersonemma2222
How do you identify the anode on a power source such as a battery? how do you identify the cathode? how are terms anion and cation?
Answers: 1

Chemistry, 22.06.2019 14:00, asanchez4292
What type of matter is made of only one kind of atom
Answers: 2
Do you know the correct answer?
Questions in other subjects:

German, 10.12.2021 01:00



Mathematics, 10.12.2021 01:00

Mathematics, 10.12.2021 01:00

History, 10.12.2021 01:00




Chemistry, 10.12.2021 01:00