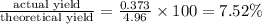Chemistry, 25.01.2021 18:00, MikeCrotch19251
2H2 (1) + O2(g) → 2H20 (g)
1. Find the limiting reactant if you start with 30.0 grams of hydrogen and 5.29 grams of oxygen.
2. The actual yield for H2O in the above reaction is 6.72 g, Determine the percent yield for the reaction
when 9.93 grams of hydrogen and excess oxygen react?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 04:00, amandasantiago2001
Which of the following ocean acidification? are the most likely side effects of a ph less than 7.0 in the ocean b. more metal salts altering the ocean chemistry c. dissolution of the shells of marine organisms d. both a & b e. all of the above.
Answers: 3


Chemistry, 22.06.2019 23:00, DESI111609
What is the average rate of the reaction between 10 and 20 s?
Answers: 1
Do you know the correct answer?
2H2 (1) + O2(g) → 2H20 (g)
1. Find the limiting reactant if you start with 30.0 grams of hydrogen a...
Questions in other subjects:


History, 10.01.2022 04:10





History, 10.01.2022 04:10




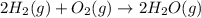
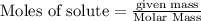
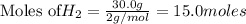
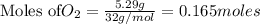
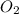 require = 2 moles of
require = 2 moles of 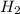
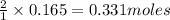 of
of 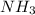
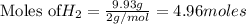
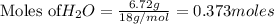
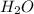
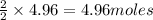 of
of