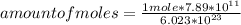Chemistry, 22.01.2021 18:50, gaboalejandro833
Calculate the numder of moles of 7.89X 10^11 atoms of Carbon.

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 23.06.2019 13:30, MalikaJones
Determine the rate law, including the values of the orders and rate law constant, for the following reaction using the experimental data provided. a + b yields products trial [a] [b] rate 1 0.30 m 0.25 m 1.2 × 10-2 m/min 2 0.30 m 0.50 m 4.8 × 10-2 m/min 3 0.60 m 0.50 m 9.6 × 10-2 m/min
Answers: 1

Chemistry, 23.06.2019 14:00, wendyyy1214
If you fill your car tire to a pressure of 32 psi (pounds per square inch) on a hot summer day when the temperature is 35°c (95°f), what is the pressure (in psi) on a cold winter day when the temperature is -15°c (5°f)? assume no gas leaks out between measurements and the volume of the tire does not change.
Answers: 1

Chemistry, 23.06.2019 18:10, rscott2649
Which of the following changes would increase the molar concentration of the products in any chemical reaction atequilibrium?
Answers: 2
Do you know the correct answer?
Calculate the numder of moles of 7.89X 10^11 atoms of Carbon....
Questions in other subjects:

Mathematics, 28.09.2019 13:30

History, 28.09.2019 13:30


Health, 28.09.2019 13:30


Mathematics, 28.09.2019 13:30



History, 28.09.2019 13:30