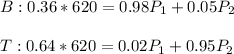Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 00:00, melidacampos12
Substance x has a fixed volume, and the attraction between its particles is strong .substance y had widely spread out particles and can be compressed what can most likely be concluded about these substances
Answers: 2

Chemistry, 22.06.2019 08:30, itzhari101
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1
Do you know the correct answer?
A mixture of 620 lbmol/h of 36 mol% benzene and the remainder toluene is separated continuously into...
Questions in other subjects:

Biology, 10.01.2021 21:40

English, 10.01.2021 21:40



Business, 10.01.2021 21:40


Mathematics, 10.01.2021 21:40

Mathematics, 10.01.2021 21:40

Mathematics, 10.01.2021 21:40

Social Studies, 10.01.2021 21:40