Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 16:10, 00015746
Predict the reactants of this chemical reaction. that is, fill in the left side of the chemical equation. be sure the equation you submit is balanced. (you can edit both sides of the equation to balance it, if you need to.) note: you are writing the molecular, and not the net ionic equation. > cacl2(aq) + h20(l)
Answers: 2

Chemistry, 22.06.2019 18:30, chinadoll24
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
Do you know the correct answer?
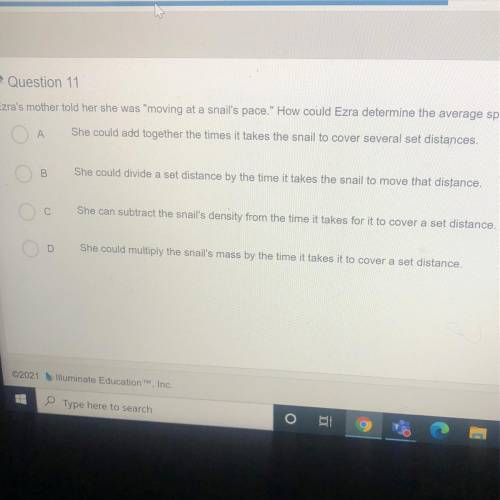
Ezra's mother told her she was "moving at a snail's pace." How could Ezra determine the average spee...
Questions in other subjects:

Mathematics, 22.04.2021 18:50

Physics, 22.04.2021 18:50






Mathematics, 22.04.2021 18:50

Mathematics, 22.04.2021 18:50

Mathematics, 22.04.2021 18:50