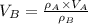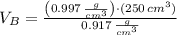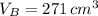Chemistry, 19.01.2021 14:00, Jazongamez1987
At 25.0 °C the density of liquid water is 0.997 g/cm3, but at -10.0 °C the density of solid water (ice) is 0.917 g/cm3. If a 250.0 mL sample of liquid water originally at 25.0 °C is frozen and cooled to -10.0 °C, what volume will the solid occupy?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 02:00, rosie20052019
Which of the following happens during cell division? (a) energy is created (b) waste is eliminated (c) carbon dioxide is released (d) damaged cells are replaced
Answers: 1

Chemistry, 22.06.2019 04:30, only1cache
When the water vapor cools it condenses select a number that represents his process on the
Answers: 3
Do you know the correct answer?
At 25.0 °C the density of liquid water is 0.997 g/cm3, but at -10.0 °C the density of solid water (i...
Questions in other subjects:

Mathematics, 12.10.2019 14:00

English, 12.10.2019 14:00




Biology, 12.10.2019 14:00


Mathematics, 12.10.2019 14:00



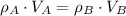 (1)
(1)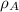 ,
, 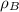 - Densities of water at 25 ºC and - 10 ºC, measured in grams per cubic centimeter.
- Densities of water at 25 ºC and - 10 ºC, measured in grams per cubic centimeter. 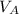 ,
, 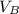 - Volume occupied by the water at 25 ºC and - 10 ºC, measured in cubic centimeters.
- Volume occupied by the water at 25 ºC and - 10 ºC, measured in cubic centimeters. 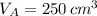 ,
, 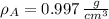 and
and 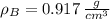 , then the volume occupied by the water at - 10 ºC is:
, then the volume occupied by the water at - 10 ºC is: