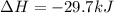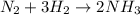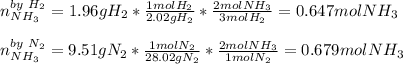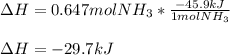Chemistry, 19.01.2021 01:00, browneyedbaby20
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9.51 g N2(g) and 1.96 g H2(g) react to produce NH3(g)

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 23:00, marissastewart533
City a and city b had two different temperatures on a particular day. on that day, four times the temperature of city a was 8â° c more than 3 times the temperature of city b. the temperature of city a minus twice the temperature of city b was â’3â° c. what was the temperature of city a and city b on that day? city a was 5â° c, and city b was 4â° c. city a was 3â° c, and city b was â’1â° c. city a was 8â° c, and city b was â’3â° c. city a was 5â° c, and city b was â’5â° c.
Answers: 2


Do you know the correct answer?
The standard molar enthalpy of formation of NH3(g) is -45.9 kJ/mol. What is the enthalpy change if 9...
Questions in other subjects:

Biology, 25.10.2019 13:43

History, 25.10.2019 13:43



Social Studies, 25.10.2019 13:43

Mathematics, 25.10.2019 13:43

History, 25.10.2019 13:43

History, 25.10.2019 13:43


Mathematics, 25.10.2019 13:43