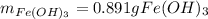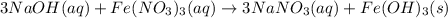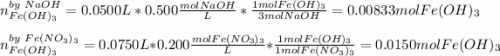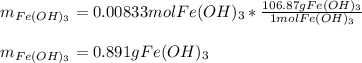Answers: 3
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 07:00, Bassoonist
How does science use models to gain a better understanding of concepts?
Answers: 1

Chemistry, 23.06.2019 08:00, IntellTanito
Pl what kind of reaction is this? nahco3 + h2o → co2 + naoh + h2o -composition -decomposition -single replacement -double replacement im leaning more toward single replacement. if im wrong can you explain whyy?
Answers: 2
Do you know the correct answer?
How many grams of iron(III) hydroxide (106.87 g/mol) will precipitate if 50.0 mL of 0.500 M sodium h...
Questions in other subjects:

Social Studies, 18.04.2020 18:48


Biology, 18.04.2020 18:48




Mathematics, 18.04.2020 18:48


English, 18.04.2020 18:48

Physics, 18.04.2020 18:48