
Chemistry, 18.01.2021 07:00, Jenniferwolf
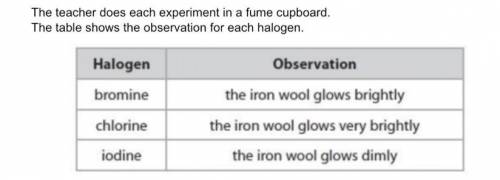
A student states that the order of reactivity cannot be found from this experiment because bromine is a liquid, chlorine is a gas and iodine is a solid at room temperature. Evaluate the student's statement.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, kylieweeks052704
Which of the following did jj thompson discover about atoms? a)an atom has an internal structure. b) atoms are tiny indivisible particles. c)electrons orbit the nucleus of an atom. d) the nucleus of an atom contains protons and neutrons.
Answers: 2

Chemistry, 22.06.2019 06:30, yolo123321
The following reaction shows sodium carbonate reacting with calcium hydroxide. na2co3 + ca(oh)2 → naoh + caco3 how many grams of naoh are produced from 20.0 grams of na2co3? (molar mass of na = 22.989 g/mol, c = 12.01 g/mol, o = 15.999 g/mol, ca = 40.078 g/mol, h = 1.008 g/mol) 12.2 grams 15.1 grams 24.4 grams 30.2 grams
Answers: 2

Chemistry, 22.06.2019 19:00, Jasoncookies23
How does kepler second law of planetary motion overthrow one of the basic beliefs of classical astronomy
Answers: 1
Do you know the correct answer?
A student states that the order of reactivity cannot be found from this experiment because bromine i...
Questions in other subjects:



History, 24.07.2019 10:30

Mathematics, 24.07.2019 10:30


Biology, 24.07.2019 10:30

Mathematics, 24.07.2019 10:30








