
Chemistry, 17.01.2021 18:40, paulitaaustin
A. Among nonpolar liquids, those with higher molar masses tend to have normal boiling points that are (higher, lower, or about the same).
b. Among compounds of approximately the same molar mass, those with greater polarities tend to have enthalpies of vaporization that are (higher, lower, or about the same).
c. Which is the only noble gas listed that is stable as a liquid at 0°C? Explain your answer using the concept of critical temperature.
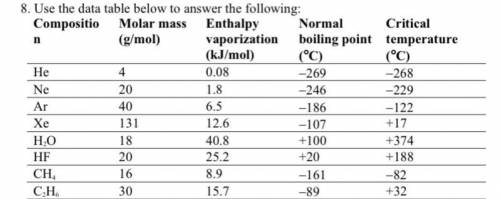

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 07:30, nayiiii1874
What three things determine the shape and size of a puddle when water is poured out onto a surface
Answers: 2

Chemistry, 22.06.2019 14:00, hammackkatelyn60
The content of manganese (mn) in steel was determined spectrophotometrically and with the use of the standard addition method. an unknown sample of mn from a digested steel sample gave an absorbance of 0.185 when analyzed spectrophotometrically. when 5.00 ml of solution containing 95.5 ppm mn was added to 50.0 ml of the unknown steel solution (digested sample), the absorbance was 0.248. calculate the concentration, in parts-per-million (ppm), of mn in the digested steel sample solution.
Answers: 3


Chemistry, 22.06.2019 19:00, Farhan54019
Which change to the system wood cause the freely-moving piston to lower?
Answers: 1
Do you know the correct answer?
A. Among nonpolar liquids, those with higher molar masses tend to have normal boiling points that ar...
Questions in other subjects:



Mathematics, 27.03.2021 23:10





Mathematics, 27.03.2021 23:10

Mathematics, 27.03.2021 23:10






