
Chemistry, 15.01.2021 01:40, rleiphart1
A hydrogen atom absorbs a photon radiation causing an electron to jump from level one to level two. If the photon/radiation has a wavelength of 5.62 x 10^-3 m to calculate the frequency of the radiation absorbed and the energy change of the electron
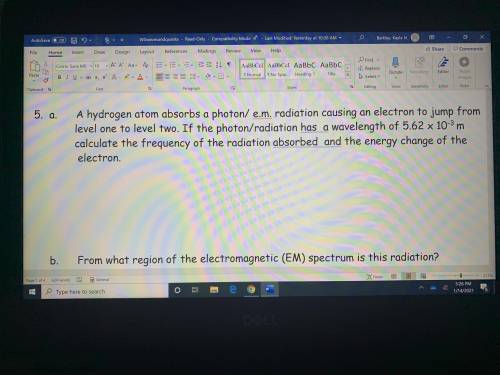

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 18:30, ElizabethF
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3


Chemistry, 22.06.2019 10:30, ashlpiriz123
Find the number of grams of hcl needed to react completely with .50 moles of magnesium.
Answers: 1
Do you know the correct answer?
A hydrogen atom absorbs a photon radiation causing an electron to jump from level one to level two....
Questions in other subjects:

History, 09.09.2020 15:01








Mathematics, 09.09.2020 15:01






