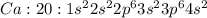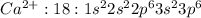Write the full electron configuration of calcium.
152 2s22p6 352 4s2
Use the electron c...

Chemistry, 14.01.2021 22:40, Randomtrashygirl
Write the full electron configuration of calcium.
152 2s22p6 352 4s2
Use the electron configuration to predict the most likely oxidation state of calcium?
(Explain your answer please)

Answers: 1
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 19:20, evansh78
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 22.06.2019 22:30, eduardoguizar8787
Which one of the following bonds would you expect to be the most polar? a) b–h b) n–h c) p–h d) al–h e) c–h
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Mathematics, 13.09.2021 19:40




History, 13.09.2021 19:40


Mathematics, 13.09.2021 19:40


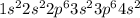 , oxidation state = +2
, oxidation state = +2