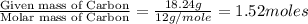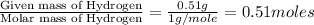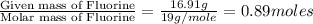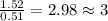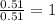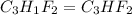Chemistry, 14.01.2021 17:00, bcarri4073
determine the empirical and molecular formula of a compound composed of 18.24 g carbon, 0.51 g hydrogen, and 16.91 g fluorine and has a molar mass 562.0 g/mol.

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 12:10, purplefish53
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 15:20, merrickrittany
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 23.06.2019 04:00, josephicarusmarrujo
Silver reacts with oxygen to produce silver oxide. (write balanced chemical equation and identify type of chemical reaction.)
Answers: 1
Do you know the correct answer?
determine the empirical and molecular formula of a compound composed of 18.24 g carbon, 0.51 g hydro...
Questions in other subjects:



History, 05.09.2019 07:10



Mathematics, 05.09.2019 07:10

History, 05.09.2019 07:10

Mathematics, 05.09.2019 07:10

English, 05.09.2019 07:10

Mathematics, 05.09.2019 07:10


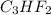 and molecular formula for the given compound is
and molecular formula for the given compound is