Chemistry, 14.01.2021 16:30, garrettbates01
Using standard heats of formation, calculate the standard enthalpy change for the following reaction. 2NH3(g) 3N2O(g)4N2(g) 3H2O(g) ANSWER: kJ Submit Answer

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 21.06.2019 22:30, shantrice1831
Using the periodic table, complete the table to describe each atom. type in your answers. a ? b? c? d? e? f?
Answers: 1

Chemistry, 22.06.2019 14:00, Killion2022
Anthracite is so hard and pure it is also referred to as a renewable resource metamorphic rock hot bituminous coal dirty fuel
Answers: 1

Chemistry, 22.06.2019 14:10, roserose3098
16. in a reaction that has reached equilibrium, a. the forward and reverse reactions are occurring at the same rate. b. the reactants and products are in equal concentrations. c. the forward reaction has gone further than the reverse reaction. d. there are equal numbers of atoms on both sides of the equation. e. a, b, and d are correct.
Answers: 2
Do you know the correct answer?
Using standard heats of formation, calculate the standard enthalpy change for the following reaction...
Questions in other subjects:

Mathematics, 23.10.2019 06:00

Mathematics, 23.10.2019 06:00

History, 23.10.2019 06:00

Biology, 23.10.2019 06:00


History, 23.10.2019 06:00

Physics, 23.10.2019 06:00



Spanish, 23.10.2019 06:00


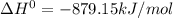
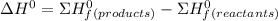
![\Delta H^{0}=3(-241.8)-[ 2(-46.2)+3(82.05)]](/tpl/images/1035/3783/3881f.png)