B. Remote sensing


Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:50, nnaomii
What is the composition, in atom percent, of an alloy that consists of 4.5 wt% pb and 95.5 wt% sn? the atomic weights for pb and sn are 207.19 g/mol and 118.71 g/mol, respectively.(a) 2.6 at% pb and 97.4 at% sn(b) 7.6 at% pb and 92.4 at% sn(c)97.4 at% pb and 2.6 at% sn(d) 92.4 at% pb and 7.6 at% sn
Answers: 2

Chemistry, 21.06.2019 22:30, officialgraciela67
Embryos of different species look very similar, which shows that the organisms share a ancestor.
Answers: 1

Chemistry, 22.06.2019 05:10, hadellolo8839
How many miles of water are produced if 5.43 mol pbo2 are consumed
Answers: 1

Chemistry, 22.06.2019 11:00, micro7909
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow. part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer. o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part. h2o is a stronger acid than oh–, so the equilibrium lies to the right. h2o is a weaker acid than oh–, so the equilibrium lies to the left. h2o is a stronger acid than oh–, so the equilibrium lies to the left. h2o is a weaker acid than oh–, so the equilibrium lies to the right. part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer. ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-( aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left. part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer. no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part. hno2 is a stronger acid than h2o, so the equilibrium lies to the right. hno2 is a weaker acid than h2o, so the equilibrium lies to the left. hno2 is a stronger acid than h2o, so the equilibrium lies to the left. hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1
Do you know the correct answer?
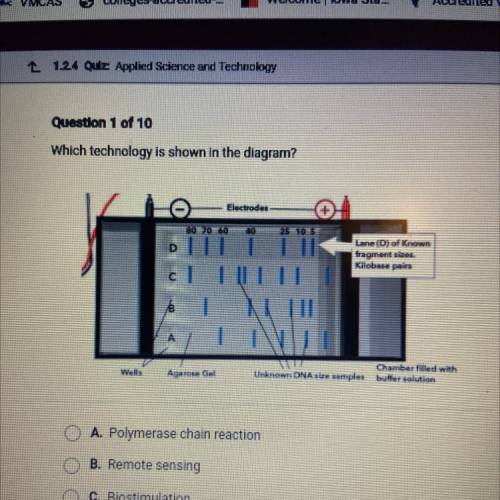
Which technology is shown in the diagram?
A. Polymerase chain reaction
B. Remote sensing
B. Remote sensing
Questions in other subjects:

English, 23.10.2020 20:00



Social Studies, 23.10.2020 20:00


English, 23.10.2020 20:00


Mathematics, 23.10.2020 20:00


Mathematics, 23.10.2020 20:00