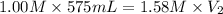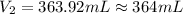Chemistry, 11.01.2021 16:10, makailaaa2
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do this by pouring out some 1.58M aqueous sodium carbonate stock solution into a graduated cylinder and diluting it with distilled water. Calculate the volume in mL of the sodium carbonate stock solution that the chemist should pour out. Round your answer to 3 significant digits.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, stellaglenn205
What reaction is taking place? 02 + c3h8 = h20 + co2
Answers: 1


Chemistry, 22.06.2019 17:30, tiffanyhmptn
Which scenario is most similar to the type of collision that gas particles have according to kinetic molecular theory
Answers: 1

Chemistry, 22.06.2019 18:00, ameliaxbowen7
Heat is the total potential energy of a substance that can be transferred. true false
Answers: 1
Do you know the correct answer?
A chemist must prepare 575.mL of 1.00M aqueous sodium carbonate Na2CO3 working solution. He'll do th...
Questions in other subjects:

English, 29.01.2021 20:40




History, 29.01.2021 20:40

Mathematics, 29.01.2021 20:40



Mathematics, 29.01.2021 20:40

Mathematics, 29.01.2021 20:40


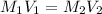
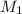 = molarity of aqueous sodium carbonate
= molarity of aqueous sodium carbonate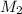 = molarity of aqueous sodium carbonate stock solution
= molarity of aqueous sodium carbonate stock solution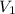 = volume of aqueous sodium carbonate
= volume of aqueous sodium carbonate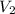 = volume of aqueous sodium carbonate stock solution
= volume of aqueous sodium carbonate stock solution