
Chemistry, 11.01.2021 05:50, abdulspy786
What mass of silver chloride can be produced from 1.01 L of a 0.196 M solution of silver nitrate? Express your answer with the appropriate units.
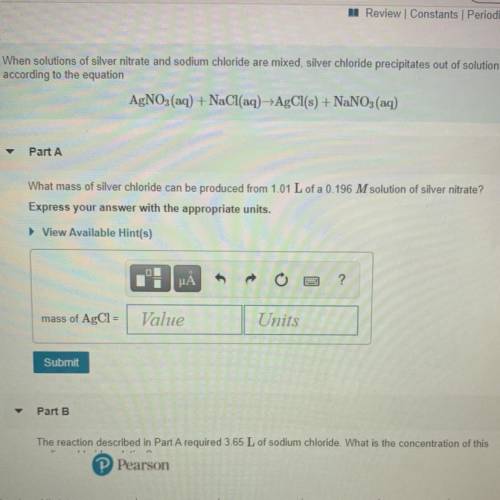

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 14:30, darkghostmist
What type of reaction fuels the processes seen here?
Answers: 2

Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1

Chemistry, 22.06.2019 19:30, dorindaramirez0531
Which liquid (h2o, h2o + soap, or h2o + salt) has the strongest cohesion and adhesion? (need now plz)
Answers: 1
Do you know the correct answer?
What mass of silver chloride can be produced from 1.01 L of a 0.196 M solution of silver nitrate?
E...
Questions in other subjects:


Geography, 08.04.2020 22:25


Mathematics, 08.04.2020 22:25

Social Studies, 08.04.2020 22:25

Mathematics, 08.04.2020 22:25

History, 08.04.2020 22:25


Social Studies, 08.04.2020 22:25

Mathematics, 08.04.2020 22:25






