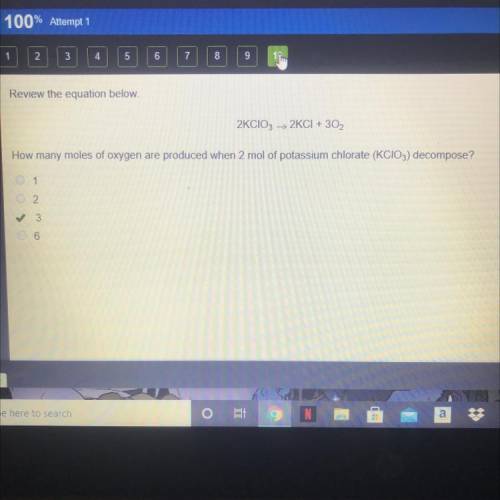
Review the equation below.
2KCIO3 > 2KCI + 3O2
How many moles of oxygen are produced...

Chemistry, 09.01.2021 22:10, tessalopezgarcia2345
Review the equation below.
2KCIO3 > 2KCI + 3O2
How many moles of oxygen are produced when 2 mol of potassium chlorate (KCIO3) decompose?
A. 1
B. 2
C. 3
D. 6
The correct answer is C on edge 2021

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 03:00, litttyyyu33411
Atrain travels 74 kilometers in 3 hours, and then 81 kilometers in 5 hours. what is its average speed?
Answers: 2

Chemistry, 22.06.2019 05:30, livigrace9004
Choose all the answers that apply. as ocean depth increases, temperature decreases temperature increases pressure increases pressure decreases salinity increases density increases
Answers: 2


Chemistry, 22.06.2019 16:10, sierram298
Amixture initially contains a, b, and c in the following concentrations: [a] = 0.300 m , [b] = 1.05 m , and [c] = 0.550 m . the following reaction occurs and equilibrium is established: a+2b⇌c at equilibrium, [a] = 0.140 m and [c] = 0.710 m . calculate the value of the equilibrium constant, kc.
Answers: 1
Do you know the correct answer?
Questions in other subjects:



Health, 30.01.2022 14:00




Mathematics, 30.01.2022 14:00

English, 30.01.2022 14:00


Mathematics, 30.01.2022 14:00






