
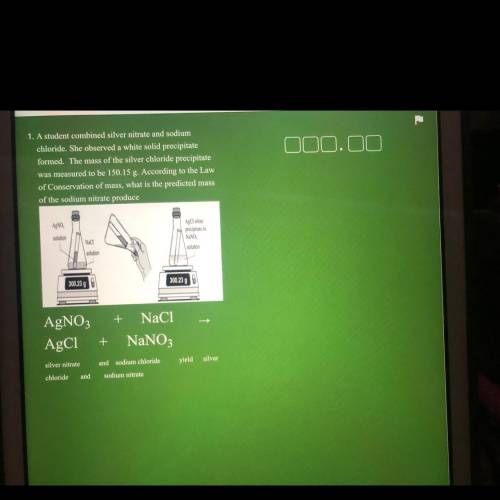
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
formed. The mass of the silver chloride precipitate
was measured to be 150.15 g. According to the Law
of Conservation of mass, what is the predicted mass
of the sodium nitrate produce
AgNO
AgCl white
precipitate in
solution
Naci
NaNO
solution
solution
300.23 g
300.23 90
+ NaCl
AgNO3
AgC1
+
NaNO3
silver nitrate
and sodium chloride
yield silver
chloride
and
sodium nitrate

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 06:00, giusto1894
24. a sports ball is inflated to an internal pressure of 1.85 atm at room temperature (25 °c). if the ball is then played with outside where the temperature is 7.5 °c, what will be the new pressure of the ball? assume the ball does not change in volume nor does any air leak from the ball a) 0.555 atm b) 1.74 atm c) 1.85 atm d) 1.97 atm
Answers: 2

Chemistry, 22.06.2019 08:00, flakko1899
An electron moved from shell n = 2 to shell n = 1. what most likely happened during the transition? a fraction of a photon was added. a photon of energy was absorbed. a fraction of a photon was removed. a photon of energy was released.
Answers: 1


Chemistry, 23.06.2019 02:40, sherlock19
How can a mixture of salt water be separated into salt and water
Answers: 1
Do you know the correct answer?
1. A student combined silver nitrate and sodium
chloride. She observed a white solid precipitate
Questions in other subjects:




Mathematics, 06.10.2020 14:01




Mathematics, 06.10.2020 14:01

Mathematics, 06.10.2020 14:01

English, 06.10.2020 14:01






