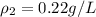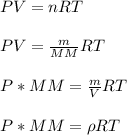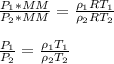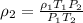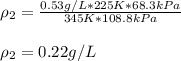Chemistry, 08.01.2021 05:10, mendezmarco2004
A sample of gas has a density of 0.53 g/L at 225 K and under a pressure of 108.8 kPa. Find the density of the gas at 345 K under a pressure of 68.3 kPa. Assuming the mass is equal to 1 gram.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 17:00, zaehairston78531
What is the nature of the ca-cl bond in a molecule of calcium chloride (cacl2) if the electronegativity value of calcium is 1.0 and that of chlorine is 3.16?
Answers: 1


Do you know the correct answer?
A sample of gas has a density of 0.53 g/L at 225 K and under a pressure of 108.8 kPa. Find the densi...
Questions in other subjects:




Mathematics, 23.07.2019 18:00



Social Studies, 23.07.2019 18:00

Biology, 23.07.2019 18:00