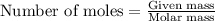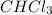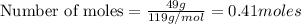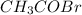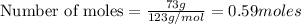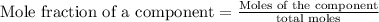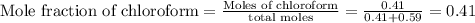Chemistry, 07.01.2021 17:40, infoneetusinghoyg22o
A solution is made by mixing 49.g of chloroform CHCl3 and 73.g of acetyl bromide CH3COBr. Calculate the mole fraction of chloroform in this solution. Round your answer to 2 significant digits.

Answers: 3
Other questions on the subject: Chemistry



Do you know the correct answer?
A solution is made by mixing 49.g of chloroform CHCl3 and 73.g of acetyl bromide CH3COBr. Calculate...
Questions in other subjects:




Chemistry, 31.07.2019 00:30

Geography, 31.07.2019 00:30

Computers and Technology, 31.07.2019 00:30

Mathematics, 31.07.2019 00:30

English, 31.07.2019 00:30


English, 31.07.2019 00:30