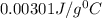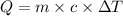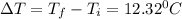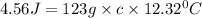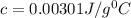Chemistry, 05.01.2021 16:30, tristanlindor5329
What is the specific heat of a 123 g substance that requires 4.56 J of heat in
order to increase its temperature by 12.32 °C?
A) 0.00301 J/g °C
B) 0.457 J/g °C
0 6910 J/g °C
D) 2.19 J/g °C
E) 0.0220 J/g°C

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 08:30, masontdavis
Draw the skeletal structures of two different molecules that are each made of 5 carbon atoms and 12 hydrogen atoms.
Answers: 1

Chemistry, 22.06.2019 12:30, hala201490
Place the elements below in order of decreasing ionization energy. aluminum(al) chlorine(cl) magnesium (mg) sulfur(s)
Answers: 1

Chemistry, 22.06.2019 12:30, gonzalesalexiaouv1bg
The missing component to the table to the right or indicated with orange letters complete the table by filling in the corresponding numbers or symbols
Answers: 3

Chemistry, 22.06.2019 13:30, yasiroarafat12
How many moles is 14.5 cm^3 of platinum? the density of platinum is 21.45 g/cm^3.
Answers: 1
Do you know the correct answer?
What is the specific heat of a 123 g substance that requires 4.56 J of heat in
order to increase it...
Questions in other subjects:

Mathematics, 14.10.2020 21:01

English, 14.10.2020 21:01

Mathematics, 14.10.2020 21:01

History, 14.10.2020 21:01




Engineering, 14.10.2020 21:01