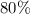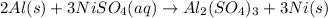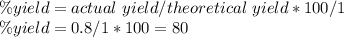Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 17:00, Estrella2209
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1


Chemistry, 22.06.2019 23:30, znewkirk4741
Imagine a small synthetic vesicle made from pure phospholipids enclosing an interior lumen containing 1 mm glucose and 1 mm sodium chloride. if the vesicle is placed in pure water, which of the following happens faster? a. na+ diffuses out. b. cl– diffuses out. c. h2o diffuses in. d. glucose diffuses out. e. sodium chloride diffuses out.
Answers: 3

Do you know the correct answer?
Aluminum metal reacts with aqueous nickel(II) sulfate to produce aqueous aluminum sulfate and nickel...
Questions in other subjects:



Chemistry, 06.11.2020 02:40




History, 06.11.2020 02:40

Mathematics, 06.11.2020 02:40


Mathematics, 06.11.2020 02:40