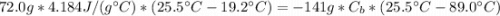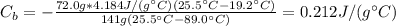Chemistry, 16.12.2020 16:50, 2025batisteajibola
A calorimeter contains 72.0 g of water at 19.2 oC. A 141 g piece of metal is heated to 89.0 oC and dropped into the water. The entire system eventually reaches 25.5 oC . What is the specific heat of the metal?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 19:30, sammysosa121832
The ph of carrots are 5.0 how it is classified a. acidic b. basic c. indicator d. neutral
Answers: 2

Chemistry, 22.06.2019 03:00, ian2006huang
Which of these would be caused by a chemical change? a) the formation of lava. b) sedimantary rock layering over time. c) metamorphic rock forming from igneous. d) metamorphic rock eroding to form sedimentary rock.
Answers: 3

Chemistry, 22.06.2019 07:30, bbyniah123
11. phosphorus-32 is radioactive and has a half life of 14 days. how much of a 124 mg sample of phosphorus-32 is present after 56 days? a) 7.75 mg b) 15.5 mg c) 31.0 mg d) 62.0 mg
Answers: 3

Chemistry, 22.06.2019 11:40, Wemaybewrong
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1
Do you know the correct answer?
A calorimeter contains 72.0 g of water at 19.2 oC. A 141 g piece of metal is heated to 89.0 oC and d...
Questions in other subjects:

Mathematics, 02.07.2019 04:00

Mathematics, 02.07.2019 04:00







English, 02.07.2019 04:00


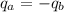

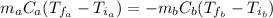
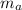 : is the water's mass = 72.0 g
: is the water's mass = 72.0 g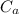 : is the specific heat of water = 4.184 J/(g°C)
: is the specific heat of water = 4.184 J/(g°C) 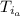 : is the initial temperature of the water = 19.2 °C
: is the initial temperature of the water = 19.2 °C 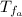 : is the final temperature of the water = 25.5 °C
: is the final temperature of the water = 25.5 °C 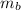 : is the metal's mass = 141 g
: is the metal's mass = 141 g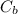 : is the specific heat of metal =?
: is the specific heat of metal =?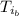 : is the initial temperature of the metal = 89.0 °C
: is the initial temperature of the metal = 89.0 °C 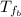 : is the final temperature of the water = 25.5 °C
: is the final temperature of the water = 25.5 °C