
Chemistry, 16.12.2020 16:50, fansofboys
Calculate the number of milliliters of 0.440 M KOH required to precipitate all of the Fe2+ ions in 187 mL of 0.692 M FeSO4 solution as Fe(OH)2. The equation for the reaction is: FeSO4(aq) + 2KOH(aq) Fe(OH)2(s) + K2SO4(aq)

Answers: 3
Other questions on the subject: Chemistry



Chemistry, 22.06.2019 14:30, davidrodriguez122001
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 22.06.2019 16:40, roderickhinton
The diagram below shows the movement of particles. what does this piece of evidence best support? the collision theory the maxwell-boltzmann distribution the effect of pressure on reaction rates the effect of temperature on reaction rates
Answers: 3
Do you know the correct answer?
Calculate the number of milliliters of 0.440 M KOH required to precipitate all of the Fe2+ ions in 1...
Questions in other subjects:

Geography, 19.03.2021 20:40


History, 19.03.2021 20:40

Mathematics, 19.03.2021 20:40

Mathematics, 19.03.2021 20:40

Mathematics, 19.03.2021 20:40




Health, 19.03.2021 20:40


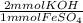 = 258.808 mmol KOH
= 258.808 mmol KOH



