
Chemistry, 14.12.2020 21:20, jaideeplalli302
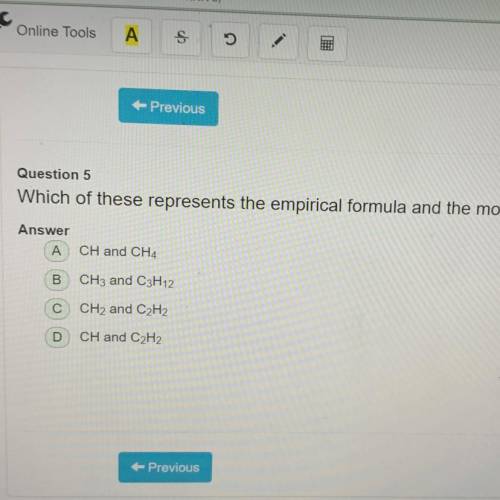
Which of these represents the empirical formula and the molecular formula for a given compound? (TEKS 8D.3)

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 23:00, poolwaterisgross
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1

Chemistry, 23.06.2019 08:00, yddlex
Suppose a pair of chemical compounds a and b can react in two different ways: a + b -> c reaction 1 gives product c. a + b -> d reaction 2 gives product d. the following facts are known about the two reactions: . reaction 1 is endothermic and reaction 2 is exothermic. if a reaction vessel is charged (filled) with a and b , then at first d is produced faster than c. use these facts to sketch a qualitative reaction energy diagram for both reactions. note: because these sketches are only qualitative, the energies don? t have to be exact. they only have to have the right relationship to each other. for example, if one energy is less than another, that fact should be clear in your sketch.
Answers: 3

Chemistry, 23.06.2019 13:30, MalikaJones
Determine the rate law, including the values of the orders and rate law constant, for the following reaction using the experimental data provided. a + b yields products trial [a] [b] rate 1 0.30 m 0.25 m 1.2 × 10-2 m/min 2 0.30 m 0.50 m 4.8 × 10-2 m/min 3 0.60 m 0.50 m 9.6 × 10-2 m/min
Answers: 1
Do you know the correct answer?
Which of these represents the empirical formula and the molecular formula for a given compound? (TEK...
Questions in other subjects:



English, 26.01.2021 19:00


English, 26.01.2021 19:00



History, 26.01.2021 19:00

Mathematics, 26.01.2021 19:00






