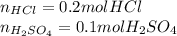Chemistry, 14.12.2020 14:00, SpiritedAway702
You are given 10ml (M) 20 Naoh solution in a conical flask and asked to titrate with (M) 20 Hcl and (M) 20 H2so4 separately. calculate the moles of Hcl and H2so4 required to neutralize the Naoh solution

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, nikejose11
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3


Chemistry, 22.06.2019 12:30, azzyla2003
Write the chemical formula for a compound that is made of an element from group 1 and an element from group 17
Answers: 1
Do you know the correct answer?
You are given 10ml (M) 20 Naoh solution in a conical flask and asked to titrate with (M) 20 Hcl and...
Questions in other subjects:

Mathematics, 20.05.2021 22:30


Mathematics, 20.05.2021 22:30


Business, 20.05.2021 22:30


Mathematics, 20.05.2021 22:30

Spanish, 20.05.2021 22:30


Mathematics, 20.05.2021 22:30