
Chemistry, 12.12.2020 17:10, jakhunter354
Indicate whether aqueous solutions of each of the following solutes will contain only ions, only molecules, or mostly molecules and a few ions:
a) NH4Cl, a strong electrolyte.
b) Ethanol, CH3-CH2-OH, a nonelectrolyte.
c) HCN, hydrocyanic acid, a weak electrolyte.

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:40, justicejesusfreak
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 13:30, justinerodriguz2878
What are the major types of a chemical compound
Answers: 2

Chemistry, 23.06.2019 00:30, natishtaylor1p8dirz
What is the chemical formula of magnesium bromide? a. mgbr2 b. mgbr c. mg2br2 d. mg2br
Answers: 3
Do you know the correct answer?
Indicate whether aqueous solutions of each of the following solutes will contain only ions, only mol...
Questions in other subjects:

Chemistry, 21.01.2021 03:50

Chemistry, 21.01.2021 03:50


Mathematics, 21.01.2021 03:50

Mathematics, 21.01.2021 03:50

Computers and Technology, 21.01.2021 03:50




Mathematics, 21.01.2021 03:50


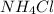 has a strong electrolyte.
has a strong electrolyte.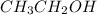 , it has a nonelectrolyte.
, it has a nonelectrolyte.



