
Chemistry, 12.12.2020 17:10, kylahwells
Calcium carbonate forms limestone, one of the most common rocks on Earth. It also forms stalactites, stalagmites, and many other types of formations found in caves. When calcium carbonate (CaCO3) is heated, it decomposes to form solid calcium oxide and carbon dioxide gas. Write the balanced equation and calculate how many liters of carbon dioxide will be produced at STP if 2.38 g of calcium carbonate reacts completely?

Answers: 2
Other questions on the subject: Chemistry




Chemistry, 23.06.2019 00:30, rose888829
The footprints of a dinosaur and the burrow of an ancient shrimp are examples of which kind of fossils
Answers: 2
Do you know the correct answer?
Calcium carbonate forms limestone, one of the most common rocks on Earth. It also forms stalactites,...
Questions in other subjects:


Mathematics, 02.07.2019 02:30

Mathematics, 02.07.2019 02:30




History, 02.07.2019 02:30

Biology, 02.07.2019 02:30



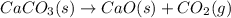 , 0.533 L
, 0.533 L 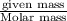
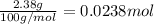
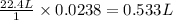 of carbon dioxide at STP
of carbon dioxide at STP



