
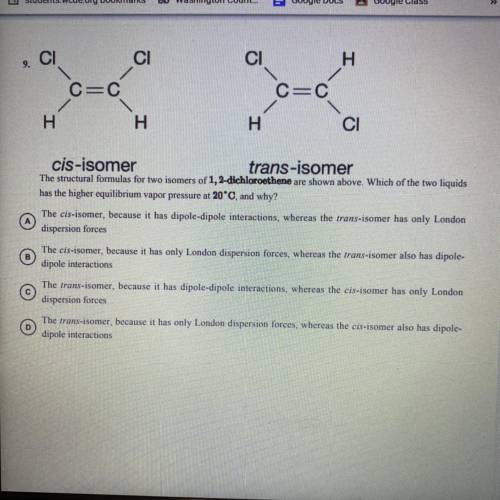
The structural formulas for two isomers of 1, 2-dichloroethene are shown above. Which of the two liquids has the higher equilibrium vapor pressure at 20 degrees Celsius and why?
A) the cis-isomer, because it has dipole-dipole interactions, whereas the trans-isomer has only 1 London dispersion forces.
B) the cis-isomer, because it has only London dispersion forces, whereas the trans-isomer also has dipole-dipole interactions
C) the trans-isomer, because it has dipole-dipole interactions, whereas the cis-isomer has only 1 London dispersion forces
D) the trans-isomer, because it has only 1 London dispersion forces, whereas the cos-isomer also has dipole-dipole interactions

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 18:50, emily9656
Which of the following is a conclusion that resulted from ernest rutherford’s scattering experiment? (will mark brainliest) a. the nucleus is negatively charged b. the atom is a dense solid and is indivisible c. the mass is conserved when atoms react chemically d. the nucleus is very small and the atom is mostly empty space
Answers: 3

Chemistry, 22.06.2019 20:10, maddie1776
Insoluble sulfide compounds are generally black in color. which of the following combinations could yield a black precipitate? check all that apply. na2s(aq)+kcl(aq) li2s(aq)+pb(no3)2(aq) pb(clo3)2(aq)+nano3(aq) agno3(aq)+kcl(aq) k2s(aq)+sn(no3)4(aq)
Answers: 1

Chemistry, 22.06.2019 21:30, sullivanjakob
Isopropyl alcohol, (ch3)2choh, is a common solvent. determine the percent by mass of hydrogen in isopropyl alcohol. a) 6.71% h b) 13.4% h c) 25.0% h d) 53.3% h
Answers: 1
Do you know the correct answer?
The structural formulas for two isomers of 1, 2-dichloroethene are shown above. Which of the two liq...
Questions in other subjects:

Mathematics, 20.11.2020 05:30



Chemistry, 20.11.2020 05:30

Mathematics, 20.11.2020 05:30


Mathematics, 20.11.2020 05:30


Mathematics, 20.11.2020 05:30






