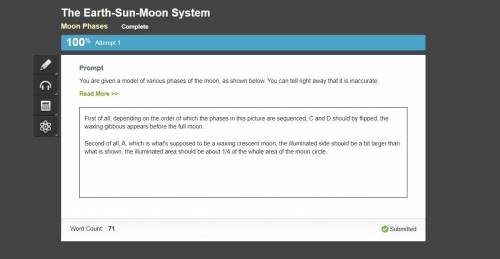
Chemistry, 10.12.2020 01:00, elleinad5656
You are given a model of various phases of the moon, as shown below. You can tell right away that it is inaccurate. Write a scientific explanation that explains the flaw in the example below.


Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, mandy9386
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1


Chemistry, 22.06.2019 09:00, yogibear5806
Look at the spectrums of a star moving towards earth and a motionless star. which of these is a correct inference that can be draw from the observation of the two spectrums? (2 points) the spectrum of a motionless star is difficult to be viewed separately using oridinary telescopes. the spectrum of a motionless star is identical to the spectrum of a star which moves towards earth. the spectrum of a star shifts towards the red region when the star moves towards earth. the spectrum of a star shifts towards the blue region when the star moves towards earth.
Answers: 2

Chemistry, 22.06.2019 11:00, micro7909
Predict the products of the following acid-base reactions, and predict whether the equilibrium lies to the left or to the right of the reaction arrow. part ao2-(aq)+h2o(l)< => express your answer as part of a chemical equation. identify all of the phases in your answer. o2-(aq)+h2o(l) < => oh-(aq)+oh-(aq)part bpredict whether the equilibrium lies to the left or to the right of the equation in previous part. h2o is a stronger acid than oh–, so the equilibrium lies to the right. h2o is a weaker acid than oh–, so the equilibrium lies to the left. h2o is a stronger acid than oh–, so the equilibrium lies to the left. h2o is a weaker acid than oh–, so the equilibrium lies to the right. part cch3cooh(aq)+hs? (aq) < => express your answer as part of a chemical equation. identify all of the phases in your answer. ch3cooh(aq)+hs-(aq) < => h2s(aq)+c2h3o2-(aq)h2s(aq)+c2h3o2-( aq)part dpredict whether the equilibrium lies to the left or to the right of the equation in previous part. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the right. ch3cooh is a weaker acid than h2s, so the equilibrium lies to the left. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the right. ch3cooh is a stronger acid than h2s, so the equilibrium lies to the left. part eno2-(aq)+h2o(l) < => express your answer as part of a chemical equation. identify all of the phases in your answer. no2-(aq)+h2o(l) < => part fpredict whether the equilibrium lies to the left or to the right of the equation in previous part. hno2 is a stronger acid than h2o, so the equilibrium lies to the right. hno2 is a weaker acid than h2o, so the equilibrium lies to the left. hno2 is a stronger acid than h2o, so the equilibrium lies to the left. hno2 is a weaker acid than h2o, so the equilibrium lies to the right.
Answers: 1
Do you know the correct answer?
You are given a model of various phases of the moon, as shown below. You can tell right away that it...
Questions in other subjects:

Mathematics, 14.09.2020 14:01

English, 14.09.2020 14:01

History, 14.09.2020 14:01

English, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

History, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01

History, 14.09.2020 14:01

Mathematics, 14.09.2020 14:01