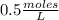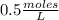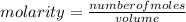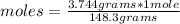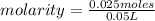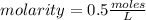The molar concentration of a solution made with 3.744 g of Mg(NO₃)₂ dissolved in enough water to make 50.0 mL of solution is 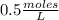
Explanation:
Molarity or Molar Concentration is the number of moles of solute that are dissolved in a certain volume.
The molarity of a solution is calculated by dividing the moles of the solute by the volume of the solution:
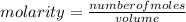
In this case:
Mg: 24.3 g/moleN: 14 g/moleO: 16 g/mole
So, the molar mass of Mg(NO₃)₂ is:
Mg(NO₃)₂= 24.3 g/mole + 2*(14 g/mole + 3*16 g/mole)= 148.3 g/mole
So, if you have 3.744 g of Mg(NO₃)₂, you can apply the following rule of three: if 148.3 grams of Mg(NO₃)₂ are present in 1 mole, 3.744 grams in how many moles are present?
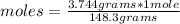
moles= 0.025
Then you have:
number of moles=0.025volume= 50 mL= 0.05 L (being 1,000 mL= 1 L)
Replacing in the definition of molarity:
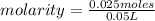
you get:
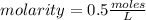
The molar concentration of a solution made with 3.744 g of Mg(NO₃)₂ dissolved in enough water to make 50.0 mL of solution is