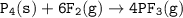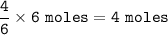Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s)...

Chemistry, 09.12.2020 17:00, tonydeanfbg8706
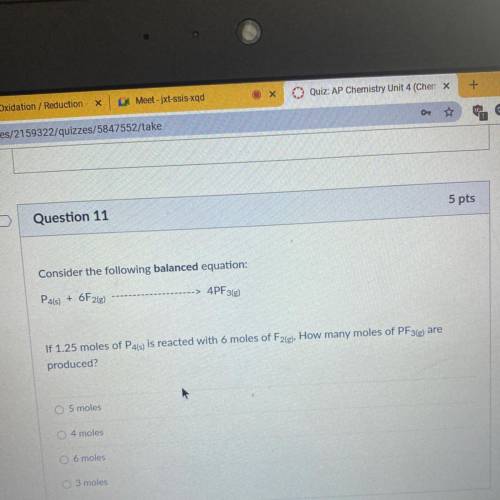
Consider the following balanced equation:
P4(s) + 6F2(e)
4PF3(g)
If 1.25 moles of P4(s) is reacted with 6 moles of F2(g), How many moles of PF3(e) are
produced?
O 5 moles
4 moles
O 6 moles
O 3 moles

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:30, Arealbot
Which statement best describes the oxidation numbers of the atoms found in magnesium chloride? a. magnesium has a 2- oxidation number and chlorine has a 1+ oxidation number. b. magnesium has a 2- oxidation number and chlorine has a 2+ oxidation number. c. magnesium has a 2+ oxidation number and chlorine has a 1- oxidation number. d. magnesium has a 1+ oxidation number and chlorine has a 1- oxidation number.
Answers: 2



Chemistry, 22.06.2019 17:00, Estrella2209
Which property of a rock remains unchanged by mechanical weathering? a. total surface area b. size and shape c. mineral composition d. sharpness
Answers: 1
Do you know the correct answer?
Questions in other subjects:


Chemistry, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30



Mathematics, 17.12.2020 20:30

Biology, 17.12.2020 20:30

Mathematics, 17.12.2020 20:30

Physics, 17.12.2020 20:30