
Chemistry, 08.12.2020 16:50, luvberries2276
200.0 mL of 3.85 M HCl is added to 100.0 mL of 4.6 M barium hydroxide. The reaction goes to completion. What is the concentration (in mol/L) of excess H+ or OH- that remains in solution?

Answers: 2
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 06:00, mapoohdoll
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1
Do you know the correct answer?
200.0 mL of 3.85 M HCl is added to 100.0 mL of 4.6 M barium hydroxide. The reaction goes to completi...
Questions in other subjects:




Mathematics, 26.03.2020 00:55


Mathematics, 26.03.2020 00:55



Mathematics, 26.03.2020 00:55


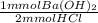 = 101.925 mmol Ba(OH)₂
= 101.925 mmol Ba(OH)₂



