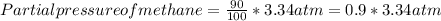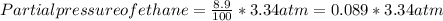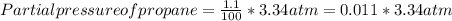Chemistry, 08.12.2020 16:30, makaylahunt
A sample of gas isolated from unrefined petroleum contains 90.0% CH4, 8.9% C2H6, and 1.1% C3H8 at a total pressure of 3.34 atm. What is the partial pressure of each component of this gas

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 22:50, ajaydonlee
Select the correct answer how does the heat content of the reaction change in the process of photosynthesis when a glucose molecule is formed? ca the value of is negative the value of qis positive the value of a remains constant the value of a decreases the value of equals zero e
Answers: 2



Chemistry, 22.06.2019 05:30, ayoismeisjjjjuan
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1
Do you know the correct answer?
A sample of gas isolated from unrefined petroleum contains 90.0% CH4, 8.9% C2H6, and 1.1% C3H8 at a...
Questions in other subjects:

Advanced Placement (AP), 02.12.2020 19:00


French, 02.12.2020 19:00




Mathematics, 02.12.2020 19:00

Spanish, 02.12.2020 19:00


Social Studies, 02.12.2020 19:00