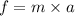Answers: 2
Other questions on the subject: Chemistry

Chemistry, 21.06.2019 16:30, KieraKimball
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 21.06.2019 17:00, kaylaamberd
Write the complete balanced equation for the reaction between lead (iv) oxide (pbo2) and water (h2o).
Answers: 1


Do you know the correct answer?
The net force on an object that is accelerating at a rate of 4 m/sec is 255 N. What is the mass of t...
Questions in other subjects:


History, 08.03.2021 21:10


Mathematics, 08.03.2021 21:10

English, 08.03.2021 21:10

Arts, 08.03.2021 21:10



Mathematics, 08.03.2021 21:10