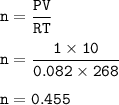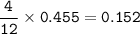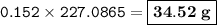Chemistry, 04.12.2020 14:00, littlemrslazy
Suppose that 10.0 L of Carbon Dioxide gas are produced by this reaction, 4C3H5N3O9 -> 12 CO2 + 10H2O + 6N2 +O2, at a temperature of -5 degrees C, and a pressure of exactly 1 atm. Calculate the mass of nitroglycerin that must have reacted in grams.

Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 10:00, micahwilkerson9495
Select all of the methods through which a drug can enter your body. injection swallowing inhalation absorption
Answers: 2


Chemistry, 22.06.2019 16:00, hjgjlgkjg
Inside a flashbulb, oxygen surrounds a thin coil of magnesium. when the flashbulb is set off, a chemical reaction takes place in which magnesium combines with oxygen to form magnesium oxide. which of the chemical equations matches the reaction above? a. mg + o2 mgo2 + energy b. 2mg + o mg2o + energy c. 2mg + o2 2mgo + energy d. mg + o mgo + energy
Answers: 1

Chemistry, 23.06.2019 06:30, nikeahbrown
Consider the heating curve of h2o and line segments a, b, and c. several changes are taking place at a, b, and c. all but one would be an appropriate description as e move through segments a, b and then c.
Answers: 3
Do you know the correct answer?
Suppose that 10.0 L of Carbon Dioxide gas are produced by this reaction, 4C3H5N3O9 -> 12 CO2 + 10...
Questions in other subjects:

Mathematics, 06.03.2020 14:24


History, 06.03.2020 14:24

English, 06.03.2020 14:24



Mathematics, 06.03.2020 14:25

History, 06.03.2020 14:25