Chemistry, 02.12.2020 17:10, BrianKeokot7700
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liquid water at 298 K. b) Calculate the standard reaction entropy for the hydrolysis of liquid water to form oxygen and hydrogen gas. c) Do your results in (a) and (b) agree with the general rule that reaction entropies are positive if there is a net formation of gas in a reaction, and negative if the is a net reduction of gas?

Answers: 1
Other questions on the subject: Chemistry


Chemistry, 22.06.2019 10:30, angemango3423
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Do you know the correct answer?
a) Calculate the standard reaction entropy for the combustion of methane to carbon dioxide and liqui...
Questions in other subjects:

Physics, 02.03.2021 03:50

Mathematics, 02.03.2021 03:50


Mathematics, 02.03.2021 03:50



Arts, 02.03.2021 03:50

Spanish, 02.03.2021 03:50

Mathematics, 02.03.2021 03:50

Biology, 02.03.2021 03:50


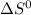 = - 242.2J/K.mol
= - 242.2J/K.mol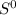 .
. 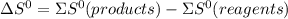
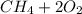 ⇒
⇒ 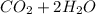
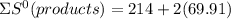 = 353.8
= 353.8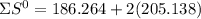 = 596.54
= 596.54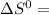 353.8 - 596.54
353.8 - 596.54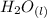 ⇒
⇒ 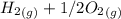
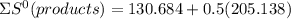 = 233.253
= 233.253