
Chemistry, 01.12.2020 20:30, Madsissabell
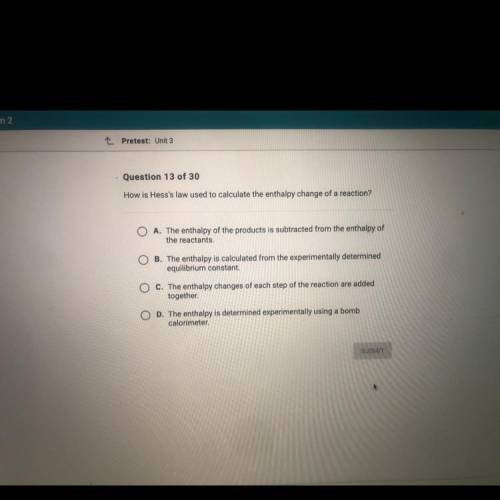
How is Hess's law used to calculate the enthalpy change of a reaction?
A. The enthalpy of the products is subtracted from the enthalpy of
the reactants,
O B. The enthalpy is calculated from the experimentally determined
equilibrium constant.
C. The enthalpy changes of each step of the reaction are added
together
O D. The enthalpy is determined experimentally using a bomb
calorimeter.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 01:30, Slycooper5959
Idon't really understand this can you me and show your work.☺☺[ chemistry b] subject [ electron transfer in lonic bonds]grade( 12)
Answers: 1


Chemistry, 22.06.2019 20:30, ashley4329
Select all the correct answers. which compounds have the empirical formula ch20? (multiple answers)a. c2h4o2b. c3h603c. ch2o2d. c5h1005e. c6h1206
Answers: 2

Chemistry, 23.06.2019 03:50, KAITLYN007
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1
Do you know the correct answer?
How is Hess's law used to calculate the enthalpy change of a reaction?
A. The enthalpy of the produ...
Questions in other subjects:


Mathematics, 27.06.2020 15:01

Mathematics, 27.06.2020 15:01



Mathematics, 27.06.2020 15:01


Biology, 27.06.2020 15:01

History, 27.06.2020 15:01

Engineering, 27.06.2020 15:01






