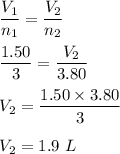Chemistry, 30.11.2020 18:00, dheydar9377
A balloon is filled to a volume of 1.50 L with 3.00 moles of gas at 25 °C. With pressure and temperature held constant, what will be the volume (in L) of the balloon if 0.80 moles of gas are added?

Answers: 2
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 05:30, fgcherubin
What happens to the atomic radius when an elctron is lost
Answers: 1

Chemistry, 22.06.2019 23:00, brianfranklin17
What is the correct lewis dot structure for arsenic?
Answers: 2

Chemistry, 23.06.2019 00:20, HernanJe6
Steam reforming of methane ( ch4) produces "synthesis gas," a mixture of carbon monoxide gas and hydrogen gas, which is the starting point for many important industrial chemical syntheses. an industrial chemist studying this reaction fills a 1.5 l flask with 3.5 atm of methane gas and 1.3 atm of water vapor at 43.0°c. he then raises the temperature, and when the mixture has come to equilibrium measures the partial pressure of carbon monoxide gas to be 1 .0 atm. calculate the pressure equilibrium constant for the steam reforming of methane at the final temperature of the mixture. round your answer to 2 significant digits.
Answers: 1

Chemistry, 23.06.2019 00:30, lareynademividp0a99r
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1
Do you know the correct answer?
A balloon is filled to a volume of 1.50 L with 3.00 moles of gas at 25 °C. With pressure and tempera...
Questions in other subjects:

Mathematics, 01.05.2021 05:00

Mathematics, 01.05.2021 05:00


Mathematics, 01.05.2021 05:00

Physics, 01.05.2021 05:00

History, 01.05.2021 05:00

Chemistry, 01.05.2021 05:00

Physics, 01.05.2021 05:00



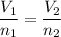 ....1)
....1)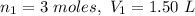
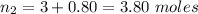
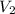 .
.