
Chemistry, 28.11.2020 07:30, xmanavongrove55
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I have to do is solve through those 2 problems but I'm not sure if I solve the fractions first then multiply that result by 100 or what. I asked my teacher which she helped a little but I don't think she understood where I was lost. If you can help I would really appreciate it. Thank you.
This was the set up question:
4. If 100.0g of nitrogen gas (N2) is reacted with 100.0g of hydrogen gas (H2) to form NH3. What is the limiting and excess reactants?
Hint: Convert grams to moles for each reactant and then convert to moles of NH3. You need your balanced equation from answer 1 to determine the mole relationship between each reactant and the product NH3. Use the periodic table to determine the molar mass of all chemical formulas. Fill in the “?” blanks below to show your work.
and in the screenshot it has everything I'm working with and the conclusions I need to draw from it, I can draw the conclusions just fine on my own but I need help solving.


Answers: 1
Other questions on the subject: Chemistry



Chemistry, 23.06.2019 01:30, babygirl091502
In what way do investigations build scientific knowledge? the results of investigations lead to questions that cannot be tested. they reflect the opinions and social values of scientists, ensuring valid information. the results of investigations lead to new questions, which lead to new investigations. they are not influenced by the research of earlier scientists, so they are able to address gaps in understanding. i
Answers: 1

Chemistry, 23.06.2019 15:30, cicilee49
In a modern periodic table, there are seven periods. a period is any horizontal row of the periodic table, and the elements in a period have consecutive atomic numbers. a group is any vertical column in the periodic table, and there are 18 such groups. groups 3–12, also known as the “b” group elements, are called transition metals. groups 1–2 and 13–18, also known as the “a” group elements, are sometimes called the main groups. metals are characterized by malleability, ductility, conductivity, and a tendency to lose electrons. main group metals are found in groups 1 and 2. nonmetallic elements fall on the right-hand side of the periodic table, that is, groups 13–18. nonmetals have the tendency to gain electrons and are generally brittle. they can be solids, liquids, or gasses at room temperature. now, label the areas of the modern periodic table using the above information. drag the appropriate labels to their respective targets.
Answers: 1
Do you know the correct answer?
25 points , hi! please look at the attachment for the question, I'm having a hard time because all I...
Questions in other subjects:

Mathematics, 11.09.2020 03:01

Business, 11.09.2020 03:01

English, 11.09.2020 03:01

English, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Social Studies, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Mathematics, 11.09.2020 03:01

Social Studies, 11.09.2020 03:01


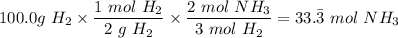
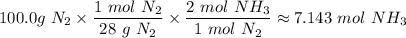
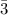 moles of NH₃)
moles of NH₃)



