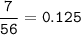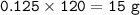Chemistry, 27.11.2020 14:00, whitakers87
Sulfur dioxide is an atmospheric pollutant.
Sulfur dioxide pollution is reduced by reacting calcium oxide with sulfur dioxide to
produce calcium sulfite.
CaO + SO2 → CaSO3
7.00 g of calcium oxide reacts with an excess of sulfur dioxide.
Relative atomic masses (Ar): O = 16 S = 32 Ca = 40 Calculate the mass of calcium sulfite produced


Answers: 3
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 11:00, RidhaH
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural. question 2 reflects a moral or social value. question 3 refers to something that can be measured. question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 11:30, chelseychew32
What is the main reason why some developing countries fear the increase the free trade policies around the world?
Answers: 2

Chemistry, 22.06.2019 14:30, villarrealc1987
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 17:10, hahahwha
Acalorimeter is to be calibrated: 51.203 g of water at 55.2 degree c is added to a calorimeter containing 49.783 g of water at 23.5c. after stirring and waiting for the system to equilibrate, the final temperature reached is 37.6 degree c. specific heat capacity of water (s = 4.18 j/g∙degree c). calculate the calorimeter constant. (smδt)warm water = -[(smδt)cold water + (calorimeterδtcold water)]
Answers: 2
Do you know the correct answer?
Sulfur dioxide is an atmospheric pollutant.
Sulfur dioxide pollution is reduced by reacting calcium...
Questions in other subjects:




Mathematics, 02.03.2021 16:50


Biology, 02.03.2021 16:50

Chemistry, 02.03.2021 16:50