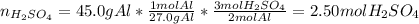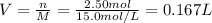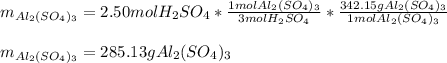Chemistry, 26.11.2020 21:50, JesusisLord2881
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)a. Determine the volume (mL) of 15.0 M sulfuric acid needed to react with 45.0 g of aluminum to produce aluminum sulfate. b. Determine the % yield if 112 g of aluminum sulfate is produced under the above conditions.

Answers: 1
Other questions on the subject: Chemistry

Chemistry, 22.06.2019 13:00, monkeyrose1999
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3


Chemistry, 22.06.2019 23:00, aly02
Need asap question 1 minerals are organic compounds. true false question 2 what vitamin can be found in foods like oranges, grapefruits, and broccoli? a. vitamin a b. vitamin k c. vitamin c d. vitamin d question 3 what are minerals? a. chemical elements that are needed for body processes. b. organic compounds that the body needs in small amounts to function properly. c. small molecules used to build proteins. d. an organic compound that is insoluble in water and includes fats. question 4 how many types of vitamins does the human body need? a. 15 b. 11 c. 13 d. 17 question 5 vitamins are a good source of energy. true false
Answers: 1

Chemistry, 22.06.2019 23:30, sweaversw9602
What are the similarities between compounds and mixtures?
Answers: 3
Do you know the correct answer?
2Al(s) + 3H2SO4(aq) → Al2(SO4)3(aq) + 3H2(g)a. Determine the volume (mL) of 15.0 M sulfuric acid nee...
Questions in other subjects:

Social Studies, 03.05.2021 21:00



Chemistry, 03.05.2021 21:00



Mathematics, 03.05.2021 21:00